Heat and temperature are related to each other, but are different concepts. Heat is the total energy of molecular motion in a substance while temperature is a measure of the average energy of molecular motion in a substance..
Besides, what is the similarities between heat and temperature?
temperature is more concerned with molecular kinetic energy . Temperature describes the average kinetic energy of molecules . HEAT deals with thermal energy . Thermal energy can be otherwise understood as the total microscopic kinetic and potential energy of a system.
Subsequently, question is, what is the difference between heat and temperature quizlet? Temperature is the average kinetic energy of the molecules (how hot or cold something feels). Heat energy is the energy created y the motion of the molecules in an object.
Subsequently, question is, what is the difference between heat and temperature grade 7?
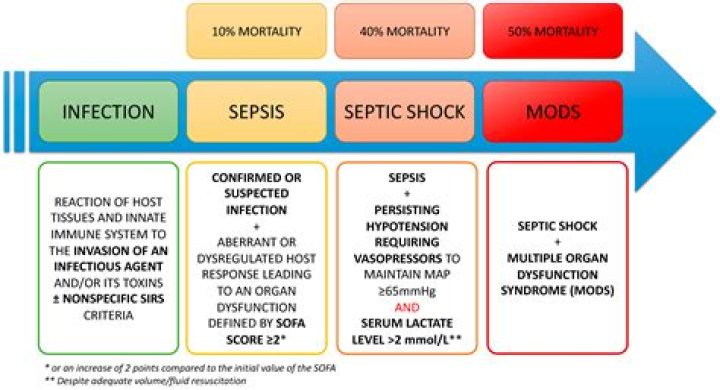
7 Heat has the ability to do work. 7 Temperature can be used only to measure the degree of heat. 8 Heat is the measure of how many molecules are in an object multiplied by how much energy each molecule possesses. 8 Temperature is related to how fast the molecules move within the object.
What is the difference between heat and energy?
The difference between heat and thermal energy is that thermal energy is not in the process of being transferred; it is not in transit, but remains as part of the internal energy of the system; heat, on the other hand, is energy in transit, i.e. energy in the process of being transferred from a hotter system towards
Related Question Answers
What is the unit of heat?
As a form of energy, heat has the unit joule (J) in the International System of Units (SI). However, in many applied fields in engineering the British thermal unit (BTU) and the calorie are often used. The standard unit for the rate of heat transferred is the watt (W), defined as one joule per second.What is difference between heat and hot?
As verbs the difference between hot and heat is that hot is to heat, or to become hot while heat is to cause an increase in temperature of an object or space; to cause something to become hot; often with "up".What are the different modes of heat transfer?
Heat can be transferred from one place to another by three methods: conduction in solids, convection of fluids (liquids or gases), and radiation through anything that will allow radiation to pass. The method used to transfer heat is usually the one that is the most efficient. How do you distinguish between temperature and heat give example?
Temperature is the degree of hotness and coldness of a body. Heat is the total kinetic energy and potential energy obtained by molecules in an object. Temperature is the average K.E of molecules in a substance. Heat flows from hot body to cold body.What are the methods of heat transfer?
Heat can travel from one place to another in three ways: Conduction, Convection and Radiation. Both conduction and convection require matter to transfer heat. If there is a temperature difference between two systems heat will always find a way to transfer from the higher to lower system.How do you define enthalpy?
Enthalpy is a thermodynamic property of a system. It is the sum of the internal energy added to the product of the pressure and volume of the system. It reflects the capacity to do non-mechanical work and the capacity to release heat. Enthalpy is denoted as H; specific enthalpy denoted as h.How is temperature and heat related?
Explanation: heat is the total energy of the motion of the molecules inside the object or particle, whereas Temperature is merely a measure of this energy. The relationship could be, the more heated an object is there higher the temperature the object will have.What are the effects of heat?
Prolonged exposure to extreme heat can cause heat exhaustion, heat cramps, heat stroke, and death, as well as exacerbate preexisting chronic conditions, such as various respiratory, cerebral, and cardiovascular diseases.What are the characteristics of heat?
All matter contains heat energy. Heat energy is the result of the movement of tiny particles called atoms, molecules or ions in solids, liquids and gases. Heat energy can be transferred from one object to another. The transfer or flow due to the difference in temperature between the two objects is called heat.How can heat be measured?
Measurement of heat is done in calories. One calorie is the amount of energy required to raise one gram of water one degree Celsius. To measure heat, you divide the change in temperature of a sample of water by the mass of the water.Does mass depend on temperature?
Increasing temperature increases vapor pressure, which speeds up outgassing and evaporation, both of which reduce the mass of the object of interest (while increasing the mass of the atmosphere). Of course, even the statement that temperature has no effect on mass is not strictly true.How does a thermometer work?
A thermometer has a glass tube sealed at both ends and is partly filled with a liquid like mercury or alcohol. As the temperature around the thermometer's bulb heats up, the liquid rises in the glass tube. When it is hot, the liquid inside the thermometer will expand and rise in the tube.What is temperature a measure of?
Temperature is a measure of the average kinetic energy of the particles of a substance. The higher the temperature of an object, the higher is its kinetic energy. Kinetic energy is a type of energy associated with motion. The units that are used to measure temperature are called degrees.What will happen if you heat a liquid to high temperatures?
When heat is added to a substance, the molecules and atoms vibrate faster. As atoms vibrate faster, the space between atoms increases. The motion and spacing of the particles determines the state of matter of the substance. The end result of increased molecular motion is that the object expands and takes up more space.What is meant by specific heat?
The specific heat is the amount of heat per unit mass required to raise the temperature by one degree Celsius. The relationship between heat and temperature change is usually expressed in the form shown below where c is the specific heat. As a result, water plays a very important role in temperature regulation.What is concept of entropy?
Entropy, the measure of a system's thermal energy per unit temperature that is unavailable for doing useful work. Because work is obtained from ordered molecular motion, the amount of entropy is also a measure of the molecular disorder, or randomness, of a system.How do we get thermal energy?
Thermal energy (also called heat energy) is produced when a rise in temperature causes atoms and molecules to move faster and collide with each other. The energy that comes from the temperature of the heated substance is called thermal energy.In what direction does heat flow?
The direction of heat flow in a small element within a solid only depends on the temperature of its neighboring elements. Heat within the solid will flow from hot to cold. It will cheerfully flow downward if it is cooler down there.What physically is heat?
Heat is a form of energy that can be transferred from one object to another or even created at the expense of the loss of other forms of energy. To review, temperature is a measure of the ability of a substance, or more generally of any physical system, to transfer heat energy to another physical system.