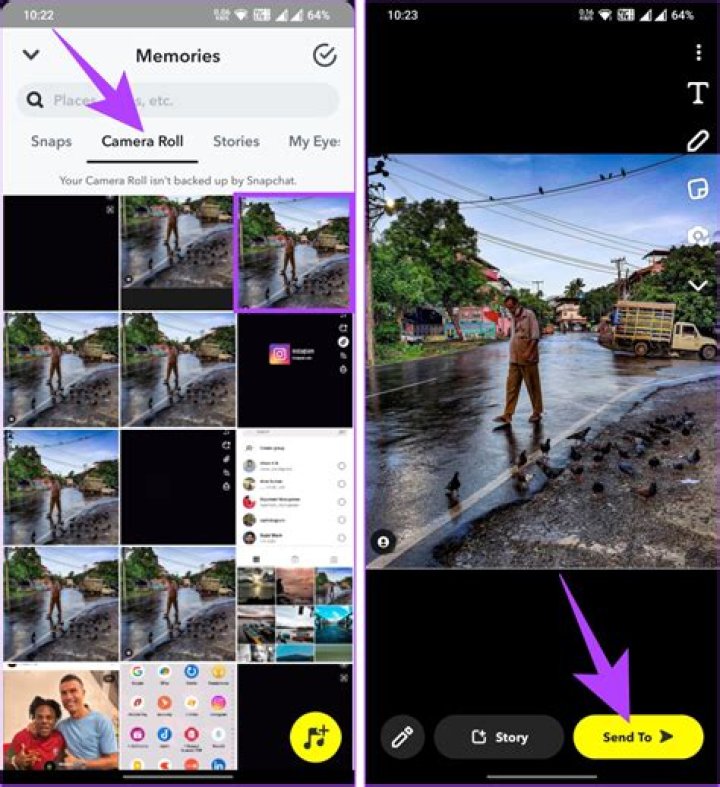
| A | B |
|---|---|
| compound | a substance made up of atoms of two or more different elements joined by chemical bonds |
| molecule | A group of atoms that are held together by chemical forces (covalent bonds); |
.
People also ask, what kind of attractive force holds two molecules together?
The hydrogen bond is one of the strongest intermolecular attractions, but weaker than a covalent or an ionic bond. Hydrogen bonds are responsible for holding together DNA, proteins, and other macromolecules.
Also, what are the different forces that hold the compounds together? Chemical bonds are forces that hold atoms together to make compounds or molecules. Chemical bonds include covalent, polar covalent, and ionic bonds. Atoms with relatively similar electronegativities share electrons between them and are connected by covalent bonds.
Also know, what holds two molecules together?
The bonds that hold atoms together to form molecules are called covalent bonds. They are pretty tough and not easily made or broken apart. It takes energy to make the bonds and energy is released when the bonds are broken.
What are the 4 types of bonds?
4 Types of Chemical Bonds
- 1Ionic bond. Ionic bonding involves a transfer of an electron, so one atom gains an electron while one atom loses an electron.
- 2Covalent bond. The most common bond in organic molecules, a covalent bond involves the sharing of electrons between two atoms.
- 3Polar bond.
What is the strongest bond?
Exception - 1/some substance form a network structure of covalent bond , like all people's are holding hand together , in that case covale Generally ionic bond is the strongest chemical bond .What is the strongest intermolecular force?
hydrogen bondingWhat are intramolecular forces?
An intramolecular force is any force that binds together the atoms making up a molecule or compound, not to be confused with intermolecular forces, which are the forces present between molecules. Chemical bonds are considered to be intramolecular forces, for example.What is Van der Waals bonding?
Van der Waals forces include attraction and repulsions between atoms, molecules, and surfaces, as well as other intermolecular forces. They differ from covalent and ionic bonding in that they are caused by correlations in the fluctuating polarizations of nearby particles (a consequence of quantum dynamics).How do you determine polarity?
Step 2: Identify each bond as either polar or nonpolar. (If the difference in electronegativity for the atoms in a bond is greater than 0.4, we consider the bond polar. If the difference in electronegativity is less than 0.4, the bond is essentially nonpolar.) If there are no polar bonds, the molecule is nonpolar.Is h2o polar or nonpolar?
A water molecule, abbreviated as H2O, is an example of a polar covalent bond. The electrons are unequally shared, with the oxygen atom spending more time with electrons than the hydrogen atoms. Since electrons spend more time with the oxygen atom, it carries a partial negative charge.What is a dipole dipole force?
Dipole-dipole forces are attractive forces between the positive end of one polar molecule and the negative end of another polar molecule. Dipole-dipole forces have strengths that range from 5 kJ to 20 kJ per mole. Polar molecules have a partial negative end and a partial positive end.How do atoms stick together?
So when two atoms are attached (bound) to one another, it's because there is an electric force holding them together. When atoms come together, they share electrons in their outer shells to form what are known as bonds. Several atoms bonded/bound together in a particular configuration is called a molecule.When might van der Waals forces be strong enough to hold molecules together?
Van der Waals forces form electrostatic bonds between molecules. Intermolecular bonds including Van der Waals bonds hold the molecules together in liquids and solids and are responsible for phenomena such as the surface tension in liquids and crystals in solids.What force holds atoms together?
In an atom there are three fundamental forces that keep atoms together. electromagnetic force, strong force, and weak force. The electromagnetic force keeps the electrons attached to the atom. The strong force keeps the protons and neutrons together in the atom.Why are Van der Waals forces weak?
Van der Waals forces also known as London Dispersion Forces are weak as they are the cause of temperorary dipole movements in the atoms. It's notable in noble gases.What is an example of van der Waals forces?
Examples of van der Waals forces include hydrogen bonding, dispersion forces, and dipole-dipole interactions.Why are covalent bonds weak?
Covalent compounds are the ones having strong intra-molecular bonds. This is because the atoms within the covalent molecules are very tightly held together. Each molecule is indeed quite separate and the force of attraction between the individual molecules in a covalent compound tends to be weak.Is water a compound?
Water OxidaneHow are van der Waals forces formed?
Search form They are weak intermolecular forces caused by attractions between very small dipoles in molecules. As the number of electrons increases so does the size of the oscillating and induced dipoles, the size of the attractive forces between the molecules, and the size of the van der Waals forces.Is Naoh an ionic compound?
Sodium oxidanide Sodium hydroxideWhat is an example of a compound?
A compound is a substance that is made up of two or more elements. Some examples of compounds include the following: water, carbon dioxide, and table salt.What are the 5 intermolecular forces?
Attractive intermolecular forces are categorized into the following types:- Hydrogen bonding.
- Ionic bonding.
- Ion–induced dipole forces.
- Ion–dipole forces.
- van der Waals forces – Keesom force, Debye force, and London dispersion force.