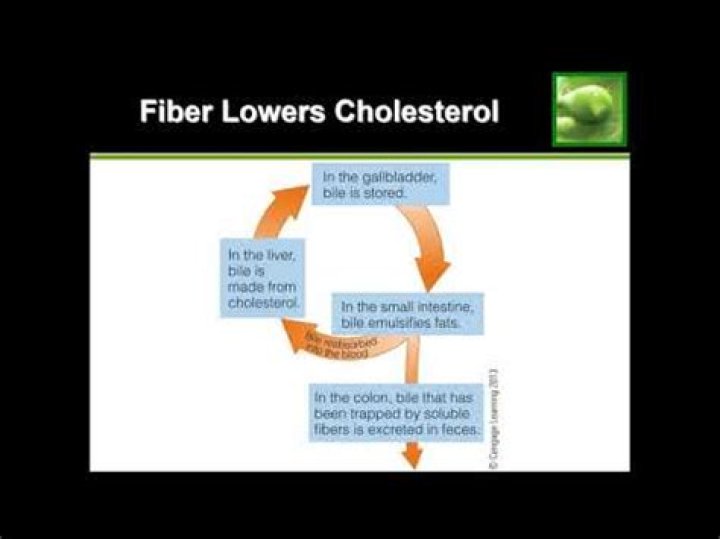
The stationary phase in paper chromatography is the strip or piece of paper that is placed in the solvent. In thin-layer chromatography the stationary phase is the thin-layer cell..
Hereof, what is stationary phase in chromatography?
Chromatography is a separation process involving two phases, one stationary and the other mobile. Typically, the stationary phase is a porous solid (e.g., glass, silica, or alumina) that is packed into a glass or metal tube or that constitutes the walls of an open-tube capillary.
Also Know, why is the stationary phase polar? In normal phase chromatography, where the stationary phase is polar, polar molecules will spend more time adsorbed on the stationary phase, while less polar ones will be carried more quickly by the non-polar mobile phase.
Secondly, what is the stationary phase in TLC?
The silica gel (or the alumina) is the stationary phase. The stationary phase for thin layer chromatography also often contains a substance which fluoresces in UV light - for reasons you will see later. The mobile phase is a suitable liquid solvent or mixture of solvents.
What is a mobile and stationary phase?
the mobile phase is the solvent that moves through the paper, carrying different substances with it. the stationary phase is contained on the paper and does not move through it.
Related Question Answers
Which of the following is not stationary phase?
Which of the following is not a stationary phase? Explanation: Solid-solid chromatography is not a stationary phase because solid-solid phase cannot provide any fluidity. Explanation: Liquid chromatography is a technique for separating ions that are dissolved in a solvent.What is meant by the term chromatography?
Definition of chromatography. : a process in which a chemical mixture carried by a liquid or gas is separated into components as a result of differential distribution of the solutes as they flow around or over a stationary liquid or solid phase.What is Rf value?
The Rf value is defined as the ratio of the distance moved by the solute (i.e. the dye or pigment under test) and the distance moved by the the solvent (known as the Solvent front) along the paper, where both distances are measured from the common Origin or Application Baseline, that is the point where the sample isHow does the stationary phase work?
Typically, the stationary phase is a porous solid (e.g., glass, silica, or alumina) that is packed into a glass or metal tube or that constitutes the walls of an open-tube capillary. The mobile phase flows through the packed bed or column.What happens during the stationary phase?
bacterial growth curve. … growth is followed by the stationary phase, in which the size of a population of bacteria remains constant, even though some cells continue to divide and others begin to die. During the stationary phase, the rate of bacterial cell growth is equal to the rate of bacterial cell death.What is the difference between stationary phase and mobile phase in chromatography?
The key difference between stationary and mobile phase is that stationary phase does not move with the sample whereas mobile phase moves with the sample. Stationary phase and mobile phase are two important terms in chromatography, which is a technique of separation and identification of the components in a mixture.What is the basic principle of chromatography?
Chromatography is based on the principle where molecules in mixture applied onto the surface or into the solid, and fluid stationary phase (stable phase) is separating from each other while moving with the aid of a mobile phase.What is meant by stationary phase and mobile phase in chromatography?
Chromatography is a method by which a mixture is separated by distributing its components between two phases. The stationary phase remains fixed in place while the mobile phase carries the components of the mixture through the medium being used.Is silica polar or nonpolar?
silica gel is very polar. so more polar material moves more slowly than nonpolar material, which feels less attraction from the silica gel. it's used in TLC and column chromatography (not paper chromatography).What is the advantage of TLC?
Advantages of TLC include rapid analysis time because many samples can be analyzed simultaneously, low solvent usage on a per-sample basis, a high degree of accuracy and precision for instrumental TLC, and sensitivity in the nanogram or picogram range.What does TLC tell you about purity?
Thin Layer Chromatography (TLC) is a separation technique requiring very little sample. It is primarily used to determine the purity of a compound. A pure solid will show only one spot on a developed TLC plate. In addition, tentative identification of the unknown compound can be made through TLC analysis.Is the stationary phase in TLC polar?
TLC uses two different phases, stationary and mobile, where the stationary phase is the very very polar silica gel and the less polar mobile phase.What happens if the TLC solvent is too polar?
If a development solvent of too high a polarity is used, all components in the mixture will move along with the solvent and no separation will be observed (Rf's will be too large). Note that the spotting solvent is simply used as a vehicle to transfer the material to be analyzed to the TLC plate.What kind of materials are used in the stationary phase for a TLC plate?
The silica gel remains the most important adsorbent for TLC separation. The kinetic properties of the silica-gel thin layer and the new TLC plates have been presented. Other materials used as stationary phase were alumina, zirconium oxide, Florisil, and ion-exchanger.What's the stationary phase of chromatography?
Chromatography is a separation process involving two phases, one stationary and the other mobile. Typically, the stationary phase is a porous solid (e.g., glass, silica, or alumina) that is packed into a glass or metal tube or that constitutes the walls of an open-tube capillary.What happens if the spots are made too large when preparing a TLC plate for development?
What happens when the spots are made too large when preparing a TLC plate for development? If the spot too large it may cause the shape of the final spot become skewed and elongates.What is polar and nonpolar?
Polar molecules occur when there is an electronegativity difference between the bonded atoms. Nonpolar molecules occur when electrons are shared equal between atoms of a diatomic molecule or when polar bonds in a larger molecule cancel each other out.What is a mobile phase and stationary phase in chromatography?
Chromatography is used to separate mixtures of substances into their components. They all have a stationary phase (a solid, or a liquid supported on a solid) and a mobile phase (a liquid or a gas). The mobile phase flows through the stationary phase and carries the components of the mixture with it.Which is more polar ferrocene or Acetylferrocene?
Ferrocene was eluted first than acetylferrocene because ferrocene is less polar than acetylferrocene. Polar compounds bind more to the stationary phase and move slower down the column. While the non-polar compounds will bind less and will elute from the column faster.