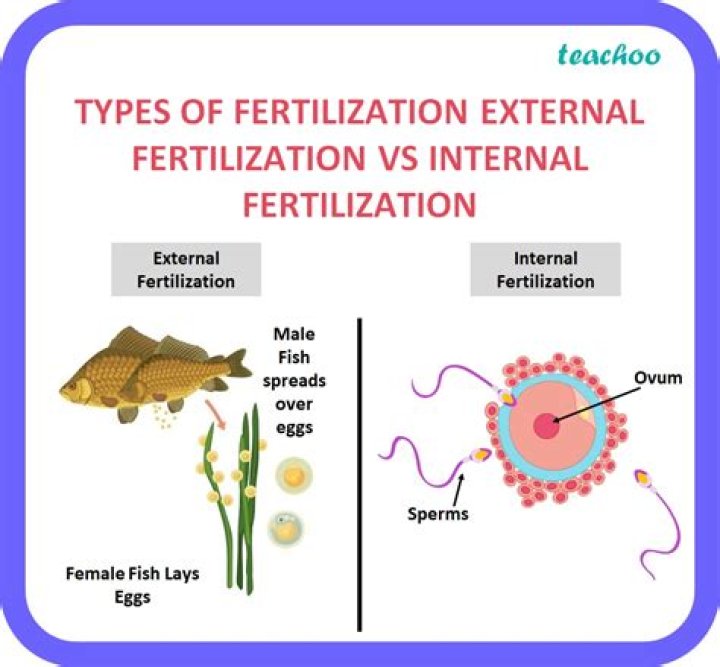
The elements are arranged in order of increasing atomic number. The horizontal rows are called periods. The vertical columns are called groups. Elements in the same group are similar to each other.
How are the elements arranged in the periodic table?
In the modern periodic table, the elements are listed in order of increasing atomic number. The atomic number is the number of protons in the nucleus of an atom. … In a periodic table arranged in order of increasing atomic number, elements having similar chemical properties naturally line up in the same column (group).
How did Mendeleev arrange the periodic table BBC Bitesize?
Mendeleev arranged the elements in order of increasing relative atomic mass . When he did this he noted that the chemical properties of the elements and their compounds showed a periodic trend .
How is the periodic table arranged GCSE?
elements are arranged in rows, called periods , in order of increasing atomic number. elements with similar properties are placed in vertical columns, called groups.How are the elements arranged in the periodic table quizlet?
In the modern periodic table, elements are arranged by increasing atomic number (number of protons). … Across a period from left to right, the elements become less metallic and more nonmetallic in their properties.
How did Dmitri Mendeleev organize the periodic table?
Mendeleev arranged the elements in order of increasing relative atomic mass . When he did this he noted that the chemical properties of the elements and their compounds showed a periodic trend .
How was the periodic table arranged before Mendeleev?
A number of other chemists before Mendeleev were investigating patterns in the properties of the elements that were known at the time. The earliest attempt to classify the elements was in 1789, when Antoine Lavoisier grouped the elements based on their properties into gases, non-metals, metals and earths.
Why are the elements arranged into groups?
The chemical elements are arranged in order of increasing atomic number. … Elements in the same group have similar chemical properties. This is because they have the same number of outer electrons and the same valency. An example of a group in the periodic table is the alkali metal group.Why is the periodic table arranged in periods?
The horizontal rows are called periods. Periods correspond to the relationship of orbitals, or likely areas in which electrons will be found, inside the outermost shell of the atom. Successive periods down the table correspond to atoms with a more electron-rich core of inner shells.
Why are elements ordered differently in the modern periodic table?Atomic number and protons After the discovery of protons , scientists realised that the atomic number of an element is the same as the number of protons in its nucleus . In the modern periodic table, the elements are arranged according to their atomic number – not their relative atomic mass .
Article first time published onWhat did Mendeleev do BBC Bitesize?
Dmitri Mendeleev He published his first periodic table of the elements in 1869. In it, he arranged the elements in order of increasing atomic weights. He also took into account the properties of the elements and their compounds .
What are the groups of the periodic table organized by quizlet?
They are organized by the atomic number from least to greatest. They are also organized using groups similar properties. What information does the periodic table contain? The periodic table provides the atomic number, atomic mass, symbol and name.
Why do you think the elements are arranged the way that they are quizlet?
Elements are arranged in order of increasing atomic number. Elements are arranged in order of increasing atomic number. Group 1 reactive. Reactivity increases down the group.
Why is the periodic table arranged the way it is quizlet?
Dmitri Mendeleev created the periodic table by arranging his elements in order of increasing atomic mass. He found out that when they were organized in this way they form a repeating pattern that periodically repeated every seven elements.
Who arranged the elements according to atomic mass and used the arrangement to predict?
Russian scientist Dmitri Mendeleev (1834-1907) is best known for developing the Periodic Table of the Elements.
Can the periodic table be arranged differently?
But there are doubts over whether the periodic table is in the best possible configuration. Just as notes can be arranged in various ways to produce music, so the essence of the relationships between the elements could be depicted differently. There is no easy way to judge which is better, or more “true”.
Why is the periodic table not arranged by atomic mass?
Assuming there were errors in atomic masses, Mendeleev placed certain elements not in order of increasing atomic mass so that they could fit into the proper groups (similar elements have similar properties) of his periodic table.
What pattern is revealed when the elements are arranged in a periodic table in order of increasing atomic number?
Moseley’s discovery was summarized as the periodic law: When elements are arranged in order of increasing atomic number, there’s a periodic pattern in their chemical and physical properties. That law led to the modern periodic table.
Why and how are elements arranged in 4th period?
Atomic structure Progressing towards increase of atomic number, the Aufbau principle causes elements of the period to put electrons onto 4s, 3d, and 4p subshells, in that order.
How are families arranged on the periodic table?
The vertical columns on the periodic table are called groups or families because of their similar chemical behavior. All the members of a family of elements have the same number of valence electrons and similar chemical properties.
How do the elements in each group differ How are they similar?
The elements in each group have the same number of electrons in the outer orbital. Those outer electrons are also called valence electrons. … Every element in the first column (group one) has one electron in its outer shell. Every element in the second column (group two) has two electrons in the outer shell.
How are the groups on the periodic table grouped?
The periodic table is organized into groups (vertical columns), periods (horizontal rows), and families (groups of elements that are similar). Elements in the same group have the same number of valence electrons. Meanwhile, elements in the same period have the same number of occupied electron shells.
Why are elements arranged according to atomic number?
The elements in the periodic table are arrange by atomic number which are equal to the number of protons. … The chemical properties are mainly dependent on the electrons, or in some cases the charge of the protons, which is independent of the number of neutrons.So,they are arranged by increasing atomic number.
How can periodic trends displayed by elements be explained?
Periodic trends displayed by the elements can be explained by structural variations of the atoms. For example, atomic size depends both on the number of protons (and how strongly these protons attract the cloud of electrons) and the number of energy levels occupied by electrons.
How would the modern periodic table be different if elements were arranged by average atomic mass instead of by atomic number?
Good question. Two reasons, actually. One is that the atomic weight of an element is the average of all its isotopes. The 12 isotope of carbon is, by definition, exactly 12, but since naturally occurring carbon contains about 1.1% ca…
Who arranged elements by atomic number?
In 1869 Russian chemist Dimitri Mendeleev started the development of the periodic table, arranging chemical elements by atomic mass. He predicted the discovery of other elements, and left spaces open in his periodic table for them.
Who organized elements into four groups?
Antoine Lavoisier, French nobleman and chemist, published the first ever known grouping of elements in a table in 1789. Lavoisier classified the elements into four categories: metals, non-metals, gases and earths.
What do elements in the same group have in common?
The elements in each group have the same number of valence electrons. As a result, elements in the same group often display similar properties and reactivity.
How many groups does the periodic table have?
Groups are numbered from 1 to 18. From left to right in the periodic table, there are two groups (1 and 2) of elements in the s-block, or hydrogen block, of the periodic table; ten groups (3 through 12) in the d-block, or transition block; and six groups (13 through 18) in the p-block, or main block.
How many elements belong to the halogen family?
The halogen elements are the six elements in Group 17 of the periodic table. Group 17 occupies the second column from the right in the periodic table and contains fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts).
What are three ways that the elements in the periodic table can be classified?
Elements can be classified as metals, metalloids, and nonmetals, or as a main-group elements, transition metals, and inner transition metals. Groups are numbered 1–18 from left to right.